Mentha arvensis
Taxonomy: Angiospermae, Lamiales, Lamiaceae, Nepetoideae, Mentheae, Menthinae, Mentha
Published: 2020-12-14
Pollen Description
Shape, Size and Aperture
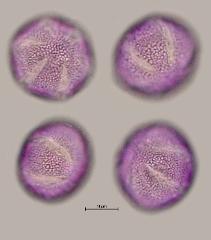
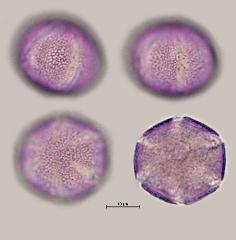
pollen unit: monad, dispersal unit and peculiarities: monad, size (pollen unit): medium-sized (26-50 µm), size of hydrated pollen (LM): 26-30 µm, shortest polar axis in equatorial view (LM): 21-25 µm, longest polar axis in equatorial view (LM): 21-25 µm, shortest diameter in equatorial or polar view (LM): 21-25 µm, longest diameter in equatorial or polar view (LM): 26-30 µm, pollen class: colpate, polarity: isopolar, P/E-ratio: oblate, shape: -, outline in polar view: elliptic, dominant orientation (LM): polar, P/E-ratio (dry pollen): prolate, shape (dry pollen): -, outline in polar view (dry pollen): elliptic, infoldings (dry pollen): aperture(s) sunken, aperture number: 6, aperture type: colpus, aperture condition: colpate, hexacolpate, aperture peculiarities: aperture membrane ornamented, stephanoaperturate
Ornamentation and Structure
LM ornamentation LM: reticulate, nexine: -, sexine: -, SEM ornamentation SEM: microreticulate, suprasculpture SEM: -, TEM tectum: semitectate, infratectum: columellate, foot layer: continuous, endexine: compact-continuous, intine: monolayered, wall peculiarities: -, supratectal element: -
Miscellaneous
pollen coatings: pollenkitt, reserves in cytoplasm: starch, cell number: 3-celled, Ubisch bodies: absent
Author(s) of diagnosis: Ulrich, Silvia; Auer, Waltraud
Pictures
Picture legend
- inflorescence(s), photographer: Ulrich, S.
- sperm cells and vegetative nucleus - dry, rehydrated (water), aceto-carmine, photographer: Ulrich, S.
- upper focus - dry, acetolyzed, unstained, photographer: Ulrich, S.
- optical section - dry, acetolyzed, unstained, photographer: Ulrich, S.
- lower focus - dry, acetolyzed, unstained, photographer: Ulrich, S.
- polar view - dry, rehydrated (water), unstained, photographer: Ulrich, S.
- pollenkitt - dry, rehydrated (water), iodine, photographer: Ulrich, S.
- pollenkitt - dry, rehydrated (water), iodine, photographer: Ulrich, S.
- hydrated pollen - fresh, glycerine, unstained, photographer: Auer, W.
- hydrated pollen - fresh, glycerine, ruthenium red, photographer: Auer, W.
- hydrated pollen - fresh, glycerine, ruthenium red, photographer: Auer, W.
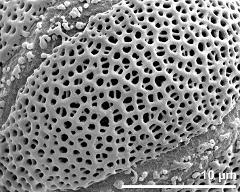
- polar view - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Ulrich, S.
- equatorial view - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Ulrich, S.
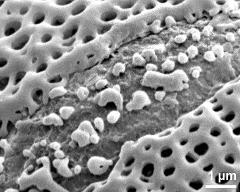
- detail of aperture - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Ulrich, S.
- exine surface - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Ulrich, S.
- exine surface - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Ulrich, S.
- polar area - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Ulrich, S.
- dry pollen grains - dry, sputter coated with gold, photographer: Ulrich, S.
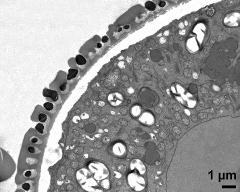
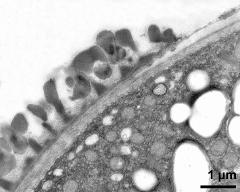
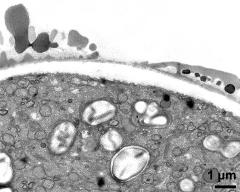
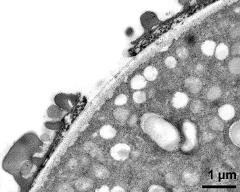
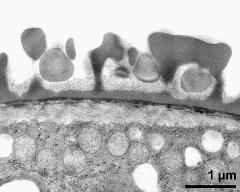
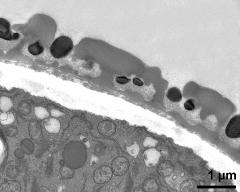
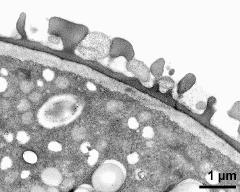
- pollen wall; interapertural area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, modified Thiéry-test, photographer: Ulrich, S.
- pollen wall at transition of aperture and interapertural area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, uranyl acetate & lead citrate, photographer: Ulrich, S.
- pollen wall; aperture area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, modified Thiéry-test, photographer: Ulrich, S.
- pollen wall at transition of aperture and interapertural area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, modified Thiéry-test, photographer: Ulrich, S.
- pollen wall; aperture area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, potassium permanganate, photographer: Ulrich, S.
- pollen wall; interapertural area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, uranyl acetate & lead citrate, photographer: Ulrich, S.
- pollen wall; interapertural area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, modified Thiéry-test, photographer: Ulrich, S.
- pollen wall; interapertural area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, potassium permanganate, photographer: Ulrich, S.
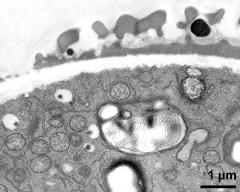
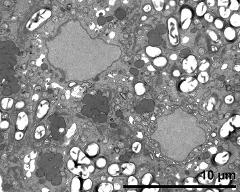
- vegetative cell and sperm cell - fresh, glutaraldehyde & osmium & potassium ferrocyanide, modified Thiéry-test, photographer: Ulrich, S.
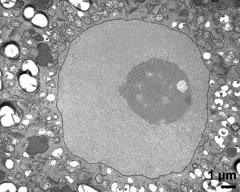
- vegetative nucleus - fresh, glutaraldehyde & osmium & potassium ferrocyanide, modified Thiéry-test, photographer: Ulrich, S.
Literature
- (1972) On the dimorphism of pollen in plants of the family Labiatae from Yakutia. Bot Zh Moscow 57: 1286-1290
- () Pollen grains in Labiatae. Madhya Bharati India II 14-16: 120-123
- (1986) Subfamilial classification of Labiatae. Systematic Botany 11: 163-185
- (1968) Labiatae: taxonomy and susceptibility to Puccinia menthae Pers. New Phytol 67: 739-743
- (1982) Pollen morphology of the West-Himalayan Labiatae. Bangladesh J Bot 11: 107-123
- (1971) A preliminary revision of Formosan Labiatae (I). Taiwania 16: 157-174
- (1989) Morphology of pollen grains of the genus Mentha L. (Lamiaceae). Acta Bot Croat 48: 161-164
- (1978) Pollen dicotyledonearum Florae Partis Europaeae USSR. Lamiaceae - Zygophyllaceae. Nauka, Akad Sci USSR, VL Komarov Inst Bot : 184 pp
- (1987) Acanthaceae, Geraniaceae, Globulariaceae, Lamiaceae, Oxalidaceae, Verbenaceae. In: Valdés B., Díez M.J., Fernández I. (eds) Atlas polínico de Andalucía occidental. Inst de Desarrollo Regional 43, Univ de Sevilla
- (1971) On the pollen morphology of Verbenaceae and a few allied taxa with special reference to taxonomy. Univ Calcutta (D Phil Thesis) : 324 pp, 18 pl, 50 figs
- (1976) Pollenmorphology of Indian Labiatae. Tribes Ocimoideae and Satureineae. J Palynology 10: 89-105
- (1956) Die Pollenkörner der Labiaten. Willdenowia 1: 617-641
- (1940) Die Pollenkörner der in Deutschland vorkommenden Labiaten. Verhandl Bot Vereins f Brandenburg 80: 21-36
- (1983) Cytopalynological studies on Indian members of Acanthaceae and Labiatae. J Palynol 19: 243-277
- (1992) Pollen morphology and taxonomy in North American Labiatae. Can J Bot 70: 975-995
- (1960) Pollen grain studies of the Labiatae of Michigan. Webbia 15: 399-415
- (1998) Preparing living pollen material for scanning electron microscopy using 2,2-dimethoxypropane (DMP) and criticalpoint drying. Biotechnic Histochem 73: 137–143
- (2005) Exkursionsflora für Österreich, Liechtenstein und Südtirol : 1180
Copyright and Citation
Cite this publication as:
Ulrich S., Auer W. 2020. Mentha arvensis. In: PalDat - A palynological database. /pub/Mentha_arvensis/304453; accessed 2026-05-14