Plantago lanceolata
Taxonomy: Angiospermae, Lamiales, Plantaginaceae, Plantago
Published: 2016-03-01
Pollen Description
Shape, Size and Aperture
pollen unit: monad, dispersal unit and peculiarities: monad, size (pollen unit): small (10-25 µm), size of hydrated pollen (LM): -, shortest polar axis in equatorial view (LM): -, longest polar axis in equatorial view (LM): -, shortest diameter in equatorial or polar view (LM): -, longest diameter in equatorial or polar view (LM): -, pollen class: porate, polarity: -, P/E-ratio: -, shape: spheroidal, outline in polar view: circular, dominant orientation (LM): -, P/E-ratio (dry pollen): -, shape (dry pollen): irregular, outline in polar view (dry pollen): irregular, infoldings (dry pollen): irregularly infolded, aperture number: >6, aperture type: porus, aperture condition: porate, pantoporate, aperture peculiarities: operculum, annulus, pantoaperturate
Ornamentation and Structure
LM ornamentation LM: verrucate, nexine: present, sexine: present, SEM ornamentation SEM: microechinate, verrucate, perforate, suprasculpture SEM: -, TEM tectum: eutectate, infratectum: granular, foot layer: discontinuous, endexine: compact-discontinuous, intine: bilayered, wall peculiarities: -, supratectal element: -
Miscellaneous
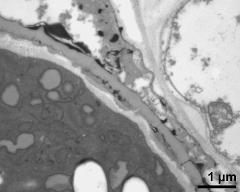
pollen coatings: absent, reserves in cytoplasm: starch, cell number: 3-celled, Ubisch bodies: present
Annotations: infratectum granular or absent; endexine only clearly visible in aperture area
Author(s) of diagnosis: Halbritter, Heidemarie; Ulrich, Silvia
Pictures
Picture legend
- habitus, photographer: Ulrich, S.
- habitus, photographer: Ulrich, S.
- inflorescence(s), photographer: Ulrich, S.
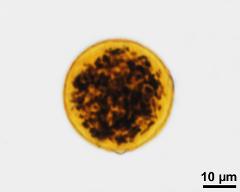
- 3-celled pollen - dry, rehydrated (water), aceto-carmine, photographer: Ulrich, S.
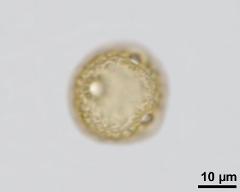
- acetolysed pollen, upper focus - dry, acetolyzed, unstained, photographer: Ulrich, S.
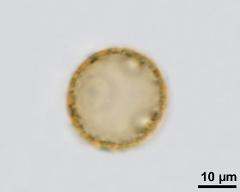
- acetolysed pollen, optical section - dry, acetolyzed, unstained, photographer: Ulrich, S.
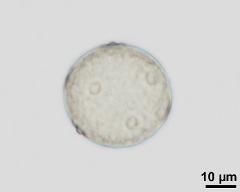
- acetolysed pollen, lower focus - dry, acetolyzed, unstained, photographer: Ulrich, S.
- hydrated pollen - dry, rehydrated (water), unstained, photographer: Ulrich, S.
- hydrated pollen - dry, rehydrated (water), unstained, photographer: Ulrich, S.
- detection of starch - dry, rehydrated (water), iodine, photographer: Ulrich, S.
- detection of starch - dry, rehydrated (water), iodine, photographer: Ulrich, S.
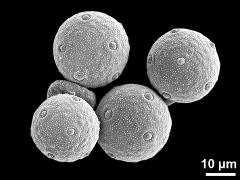
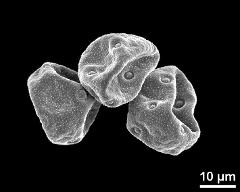
- hydrated pollen grains - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Halbritter, H.
- hydrated pollen grain - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Halbritter, H.
- hydrated pollen - dry, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Ulrich, S.
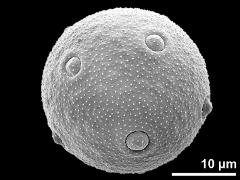
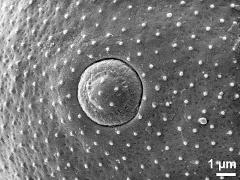
- aperture - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Halbritter, H.
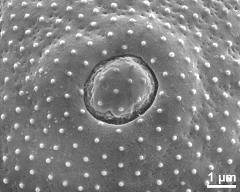
- aperture - dry, rehydration (water) & critical point dried & sputter coated with gold, photographer: Ulrich, S.
- aperture - dry, rehydration (water) & critical point dried & sputter coated with gold, photographer: Ulrich, S.
- aperture - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Halbritter, H.
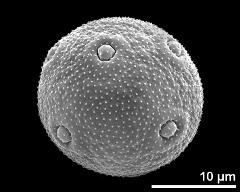
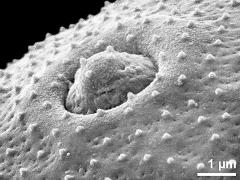
- exine surface - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Halbritter, H.
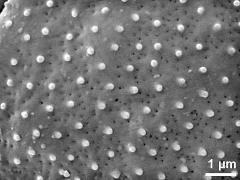
- dry pollen grains - dry, sputter coated with gold, photographer: Halbritter, H.
- dry pollen - dry, sputter coated with gold, photographer: Ulrich, S.
- Ubisch bodies on inner anther wall - fresh, rehydrated (water) & critical point dried & sputter coated with gold, photographer: Halbritter, H.
- Ubisch bodies on inner anther wall - dry, rehydration (water) & critical point dried & sputter coated with gold, photographer: Ulrich, S.
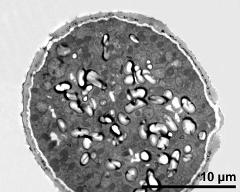
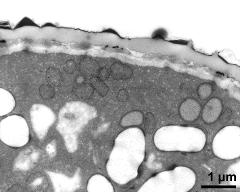
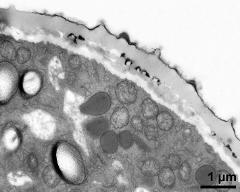
- cross section of pollen grain - fresh, glutaraldehyde & osmium & potassium ferrocyanide, modified Thiéry-test, photographer: Ulrich, S.
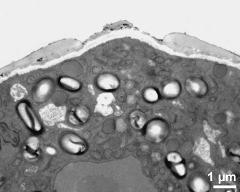
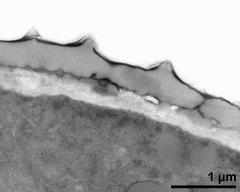
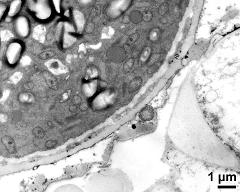
- pollen wall, aperture area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, modified Thiéry-test, photographer: Ulrich, S.
- pollen wall, aperture area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, uranyl acetate & lead citrate, photographer: Ulrich, S.
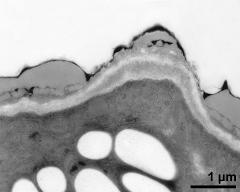
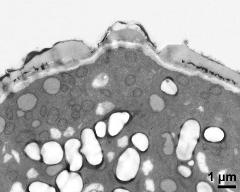
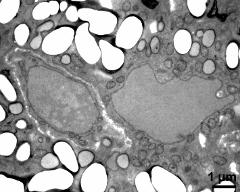
- pollen wall, interapertural area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, lipid-test, photographer: Ulrich, S.
- pollen wall, interapertural area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, uranyl acetate & lead citrate, photographer: Ulrich, S.
- pollen wall, aperture area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, modified Thiéry-test, photographer: Ulrich, S.
- pollen wall, aperture area, pollenkitt - fresh, glutaraldehyde & osmium & potassium ferrocyanide, lipid-test, photographer: Ulrich, S.
- pollen wall, aperture area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, potassium permanganate, photographer: Ulrich, S.
- pollen wall, interapertural area - fresh, glutaraldehyde & osmium & potassium ferrocyanide, modified Thiéry-test, photographer: Ulrich, S.
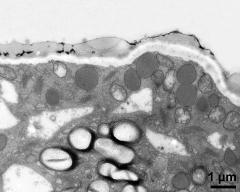
- pollen wall, Ubisch bodies on loculus wall - fresh, glutaraldehyde & osmium & potassium ferrocyanide, lipid-test, photographer: Ulrich, S.
- pollen wall, interapertural area, pollenkitt, Ubisch bodies - fresh, glutaraldehyde & osmium & potassium ferrocyanide, lipid-test, photographer: Ulrich, S.
- cytoplasma, vegetative nucleus, sperm cell, starch - fresh, glutaraldehyde & osmium & potassium ferrocyanide, modified Thiéry-test, photographer: Ulrich, S.
Literature
- (1977) Plantaginaceae. Rev Palaeobot Palynol 24: 129-154
- (1978) Entwicklungstendenzen blütenökologischer Merkmale bei Plantago. Flora 167: 41-56
- (1976) Anthecological relations between reputedly anemophilous flowers and syrphid flies. II. Plantago media L. Acta Bot Neerl 25: 205-211
- (1978) Evolutionary aspects of wind pollination in the genus Plantago (Plantaginaceae). New Phytol 81: 449-458
- (1984) The significance of biotic pollination in a nominally anemophilous plant: Plantago lanceolata. Proc Kon Ned Akad Wet, ser C 87: 95-119
- (1976) Anthecological relations between reputedly anemophilous flowers and syrphid flies. I. The possible role of syrphid flies as pollinators of Plantago. Tijdschr Entom 119: 15-31
- (1988) Palynological study of the genus Plantago in the Iberian Peninsula. Grana 27: 1-15
- (1998) Preparing living pollen material for scanning electron microscopy using 2,2-dimethoxypropane (DMP) and criticalpoint drying. Biotechnic Histochem 73: 137–143
- (2005) Exkursionsflora für Österreich, Liechtenstein und Südtirol : 1180
Copyright and Citation
Cite this publication as:
Halbritter H., Ulrich S. 2016. Plantago lanceolata. In: PalDat - A palynological database. /pub/Plantago_lanceolata/300502; accessed 2026-05-04